Glenohumeral joint arthritis is commonly accompanied by stiffness related to contracture and adhesions involving the glenohumeral capsule, the cuff muscles, and the nonarticular humeroscapular motion interface.
Causes and complications of roughness
Weakness of the cuff muscles results from disuse or fiber failure. Instability patterns may also complicate glenohumeral roughness, such as the posterior subluxation characteristic of degenerative joint disease and capsulorrhaphy arthropathy, or the superior subluxation characteristic of cuff tear arthropathy. There is a lot about these conditions that needs management!
Nonoperative management possibilities
Because glenohumeral roughness is usually of insidious onset and chronic duration, there is ample opportunity to try nonoperative management. In many instances of glenohumeral arthritis, the mechanics of the shoulder can be improved by a program of patient-conducted, gentle, range-of-motion and strengthening exercises (see Home Exercises for the Rough Shoulder and Figure 1). It is important that vigorous torques and forces not be applied in an attempt to regain motion because of the concern for causing obligate translation and accelerated wear. Nonsteriodal antiinflammatory medication and mild analgesics may be useful adjuncts.
Surgery is considered for well-informed, well-motivated, cooperative, sufficiently healthy, and socially supported patients with refractory and functionally significant glenohumeral roughness.
Surgical treatment
Surgical options
Surgical reconstruction offers the potential to optimize capsular laxity and muscle mechanics, as well as the smoothness, size, shape, and orientation of the joint surface. While prosthetic arthroplasty is the primary surgical option to be considered when major pain and functional loss result from glenohumeral arthritis, other surgical alternatives have been described in the management of arthritis. Ian Kelly (Kelly 1990; Kelly 1994) Bennett and Gerber (Bennett and Gerber 1994) and Thomas et al (Thomas Amstutz and Cracchiolo 1991) have recently reviewed some of the surgical options for surgical management of the rheumatoid shoulder.
Synovectomy in Inflammatory Arthritis
Inflammatory arthropathies, such as Rheumatoid Arthritis (RA), are characterized by synovial tissue hyperplasia—an overgrowth of the joint lining that leads to chronic pain and swelling.
Historical Perspective
A foundational 1943 study by Smith-Peterson, Aufranc, and Larson remains a benchmark for understanding surgical intervention in the pre-prosthetic era. Their insights established several key principles:
Systemic Nature: Surgery on a specific joint is unlikely to alter the overall long-term progression of systemic disease.
Conservative Approach: Surgical intervention is not always necessary or even desirable for every patient.
Symptom Management: For patients with severe, persistent shoulder symptoms, procedures such as synovectomy, bursectomy, and acromioplasty were found to significantly alleviate pain and restore limb function.
Clinical Indications
Synovectomy is generally reserved for patients suffering from chronic refractory synovitis—inflammation that fails to respond to medical management.
Diagnostic Signs:
Physical Exam: A characteristically "boggy" or enlarged feeling upon palpation of the shoulder.
Pathology: This typically indicates significant primary bursal hypertrophy or may suggest a rotator cuff tear with associated synovial extension.
Arthroplasty
Historical review
In 1893, one of the first prosthetic shoulder replacements was performed by the French surgeon Pean. (Lugli 1978) Pean was the subject of a painting called "Une Operation de Tracheotomie" by Henri de Toulouse-Lautrec. (Bankes and Emery 1995) A platinum and rubber total joint and proximal humeral implant were fashioned for him by J. Porter Micheals, a dentist from Paris, and inserted by Pean in a 37 year old baker after his tuberculous arthritis had been debrided. The patient gained increased strength and range of the arm. However, the infection recurred. After one of the first Xray machines documented an overwhelming reactive process, the prosthesis was removed two years after implantation.
In 1953, Neer presented the option of replacement of a fractured humeral head with a Vitallium prosthesis. (Neer, Brown, and McLaughlin 1953) Use of this prosthesis was next applied to patients with irregular articular surfaces as a result of fracturing and osteonecrosis. (Neer 1955) In 1971 and 1974, Neer described the results of the use of this proximal humeral implant for patients with rheumatoid arthritis and osteoarthritis of the glenohumeral joint. (Neer 1971; Neer 1974) In these articles Neer also described the use of a high density polyethylene glenoid in the management of osteoarthritis of the glenohumeral joint. Also in 1974, Kenmore and associates published a brief article reporting on the development of a polyethylene glenoid liner for use with a Neer humeral replacement in the treatment of degenerative joint disease of the shoulder. (Kenmore, MacCartee, and Vitek 1974; Neer Watson, and Stanton 1982) Other early descriptions of shoulder arthroplasty components include prostheses of Vitallium, as reported by Krueger (Krueger 1951), and acrylic, as reported by Richard and Rene Judet. (Richards Judet and Rene 1952)
The initial Neer prosthesis had three sizes and two more were added. In the early 1970s, the implant was redesigned to better use the alternative of cement fixation and the articular portion was made spherical. (Neer 1974) This implant proved its versatility over time. It was initially used for acute fractures but subsequently has been shown to be effective for the care of patients with chronic fracture problems (Hawkins, Neer, Pianta, et al. 1987; Pritchett and Clark 1987; Rowe and Zarins 1982; Tanner and Cofield 1983), osteoarthritis (Neer 1974; Zuckerman and Cofield 1986), rheumatoid arthritis (Neer 1971; Zuckerman and Cofield 1986), osteonecrosis (Cruess 1976; Cruess 1985; Rutherford and Cofield 1987), and a variety of the more rare forms of disease affecting the shoulder joint. A number of other shoulder implant systems include a metallic humeral component that can be used without a glenoid replacement. (Amstutz 1982; Amstutz Sew Hoy Clarke 1981; Amstutz Thomas Kabo et al. 1988; Bateman 1978; Cofield 1986; Cofield 1987; Gristina Romano Kammire et al. 1987; Gristina Webb and Carter 1985) Bipolar implants have been described as well. (Lee and Niemann 1994; Swanson 1984; Swanson deGroot Maupin et al. 1986; Swanson deGroot Swanson Sattel et al. 1989)
Some authors suggested a cup arthroplasty might be a satisfactory alternative for prosthetic shoulder surgery. (Jonsson 1988; Jonsson Egund Kelly et al. 1986). Initially, hip cups were used (Steffee and Moore 1984) and then cups were manufactured specifically for the shoulder. (Steffee and Moore 1984) Rydholm and Sjogren (Rydholm and Sjogren 1993) described a surface replacement for the humeral head. At an average of 4.2 years after surgery, 72 rheumatoid shoulders demonstrated substantial improvement. 25% of the cups were loose at followup. However, neither cup loosening, nor proximal migration of the humerus, nor central glenoid wear apparently affected the clinical result.
A number of plastics and other softer materials have been used as implants. Swanson designed an all-silicon rubber humeral head implant in extension of the concept of flexible implants as an adjunct to resection arthroplasty. (Swanson 1973) Apparently, this design was used only on rare occasions and no results are available for a series of patients. Varian reported on a clinical trial of the use of a Silastic cup in patients with rheumatoid arthritis of the shoulder. (Varian 1980) Early results were promising, but another series by Spencer and Skirving described a number of complications and the authors recommended restricted use ofthe device. (Spencer and Skirving 1986)
Isoelastic shoulder implants have been used in Europe. (Burri 1985; Cockx Claes Hoogmartens et al. 1983; Tonino and van de Werf 1985)
After these pioneering efforts, many additional shoulder prostheses were constructed. Some mirrored the implants used by Neer, including the St. Georg (Engelbrecht and Stellbrink 1976), the Bechtol (Bechtol 1976) prostheses, the DANA (Thomas Amstutz and Cracchiolo 1991), the Cofield (Cofield 1986; Cofield 1987), and the Monospherical (Gristina Romano Kammire et al. 1987; Gristina Webb and Carter 1985). Other designs included a captive ball-in-socket unit to replace the stabilizing functions of the rotator cuff and shoulder capsule (Beddow and Elloy 1977; Beddow and Elloy 1982; Buechel Pappas and DePalma 1978; Cofield and Stauffer 1977; Coughlin Morris and West 1979; Engelbrecht and Heinert 1987; Fenlin 1975; Gerard Leblanc and Rousseau 1973; Gristina and Webb 1982; Kessel and Bayley 1982; Kolbel and Friedebold 1975; Kolbel Rohlmann and Bergmann 1982; Lettin Copeland and Scales 1982; Post 1987; Post 1988; Post and Haskell 1978; Post Haskell and Jablon 1980; Post Jablon Miller et al. 1979; Reeves Jobbins Dowson et al. 1974; Wheble and Skorecki 1977; Zippel 1975) Of these, many included complex and extensive attachments to the scapula by cementing within the glenoid and by stems, wedges, a screw, and bolted flanges. Some designs reversed the ball and socket configuration--attaching the ball part of the implant to the glenoid. (Reeves Jobbins Dowson et al. 1974) Others incorporated two ball-in-socket units. (Buechel Pappas and DePalma 1978; Gristina and Webb 1982) Engelbrecht et al suggested a hemiarthroplasty with modification of the glenoid by osteotomy and bone graft to buttress the humeral prosthesis. (Engelbrecht and Heinert 1987) Burkhead and Hutton(Burkhead and Hutton 1995) performed biological resurfacing of the glenoid in association with humeral hemiarthroplasty.
Some designs had a hood on the glenoid component in an attempt to prevent upward humeral subluxation associated with rotator cuff weakness or absence. (Amstutz 1982; Amstutz Sew Hoy and Clarke 1981; Amstutz Thomas Kabo et al. 1988; Engelbrecht and Stellbrink 1976; Mazas and de la Caffiniere 1982; McElwain and English 1987; Neer Watson and Stanton 1982) Implants could be classified as anatomical, semiconstrained (hooded glenoid), or constrained (ball in socket).
The nonretentive prosthesis of Mazas and Caffiniére also included a superiorly placed hood on the glenoid component. Of 38 shoulders operated, 9 developed instability and 14 shoulders remained stiff after surgery. (Mazas and de la Caffiniere 1982) A third early system including a hooded component was the English-Macnab. This system also has a nonhooded glenoid implant and incorporates porous ingrowth surfaces on the glenoid component and the humeral stem. (Faludi and Weiland 1983; McElwain and English 1987)
The Neer system originally included 200 per cent and 600 per cent enlarged glenoids. These components were used in only 12 of 273 shoulders reported (Neer Watson and Stanton 1982), suggesting that the need for them was quite uncommon. The Dana total shoulder arthroplasty includes a semiconstrained hooded component designed to enhance stability in shoulders with irreparable rotator cuff tears. (Amstutz 1982) The Monospherical total shoulder also incorporated a slight hood on the glenoid component imparting somewhat greater stability to the articulation. (Fukuda Chen Cofield et al. 1988; Gristina Romano Kammire et al. 1987) Laurence (Laurence 1991) has described a snap fit prosthesis for arthroplasty in the cuff deficient shoulder; the cup is secured to the glenoid and acromion.
Neer defined many of the challenges of shoulder reconstruction, including the management of malversion of the glenoid, cementing the glenoid, and proximal humeral deficiency. (Neer Watson and Stanton 1982) He expanded the diagnoses that could be managed by prosthetic shoulder reconstruction. Cofield also pioneered the extended application of shoulder arthroplasty with an expanded implant system. (Cofield 1990) Pearl and Lippitt(Pearl and Lippitt 1994) and Collins et al (Collins Harryman Lippitt et al. 1991) have recently outlined many of the elements of modern arthroplasty technique.
The 1980's saw the advent of a number of modular humeral component designs trying to accommodate the variations in humeral anatomy and space available for the joint and humeral medullary canal diameters. On the glenoid side, some designs offered cementless fixation using screws and porous coatings on metal backing to the polyethylene. In the 1990's, increased emphasis is being placed on restoring normal kinematics with anatomical location and orientation of the humeral and glenoid joint surfaces, advanced soft tissue balancing methods, and physiological stabilization of the joint. Zuckerman and Cuomo have recently provided a review of the indications and preoperative planning for glenohumeral arthroplasty. (Zuckerman and Cuomo 1993) Brems has conducted a review of the evolution of the glenoid component. (Brems 1993) Rodosky and Bigliani have reviewed the indications for glenoid resurfacing. (Rodosky and Bigliani 1996)
Unfortunately the results of most of the tens of thousands of surgeries performed for glenohumeral arthritis are not available. This is due in large part to the fact that most outcome systems are too burdensome for most of the surgeons carrying out shoulder reconstructions. Over the 20 years since the advent of shoulder arthroplasty, the results of the procedure have been published for less than 2000 cases performed in this country; an estimated 5% of the total. In that most of the published reports come from centers where relatively large numbers of these procedures are performed, it would be of immense interest to know to what degree the results of the other 95% were similar.
An important recent advance is that simple and practical systems are now available by which surgeons can easily document the status of their patients before and sequentially after shoulder arthroplasty. (Lippitt Harryman and Matsen 1993; Matsen 1996; Matsen Smith DeBartolo et al. 1996; Richards An Bigliani et al. 1994; Ziegler DeBartolo and Matsen 1995) This documentation of treatment effectiveness will permit the comparison of different management approaches for defined groups of patients. If data on over 6000 shoulder arthroplasties being performed each year can be gathered, analyzed, and compared, shoulder surgeons will be in a powerful position to understand, and to progressively improve, the effectiveness of the care they offer their patients.
Milne and Gartsman (Milne and Gartsman 1994) recently reviewed the costs of arthroplasty in 1992-3 in a Houston Texas private practice setting. They found the average for hemiarthroplasty was $15,656 and for total shoulder arthroplasty $16,606. Of this figure, 20% was for the surgeon, 75% for the hospital, 3% for the anesthesia, and 2% for consultations. 4% of the patients were on workers compensation, 43% were private pay, and 53% were on Medicare. The length of stay averaged 5 days with a range from 3-14 days.
Indications for surgery
Glenohumeral arthroplasty is a technically demanding and powerful tool for the reconstruction of the arthritic shoulder. Shoulder arthroplasty is indicated when the following conditions are met:
- Substantial disability of the shoulder exists and is clearly related to loss of the normal glenohumeral articulation.
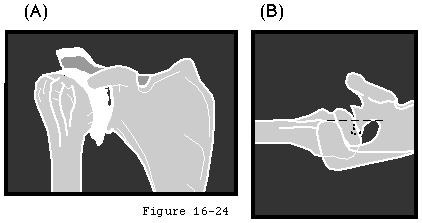
It is useful to document the both the disability and the glenohumeral destruction using standardized tools such as the Simple Shoulder Test, the SF 36, and defined radiographic views (see figures 2 and 3). - The anatomy of the shoulder is amenable to reconstruction using shoulder arthroplasty
i.e. there is sufficient bone stock, muscle strength, and tendon integrity to provide for a functional and robust reconstruction. In certain situations the presence of anatomic deficiencies may favor a hemiarthroplasty as opposed to a total shoulder. For example when there is insufficient glenoid bone to support a glenoid component or when the humeral head is fixed in a superiorly displaced position relative to the glenoid, as in cuff tear arthropathy. - The patient is committed to the success of the procedure, has no contraindications, understands the limitations of a shoulder prosthesis, and has sufficient social support for the postoperative period.
The ideal patient for a prosthetic arthroplasty has a positive attitude coupled with the understanding that a shoulder arthroplasty is not meant to be used for heavy or jerky pushing, pulling, or lifting, nor for overhead work. Active or recent infection, absent deltoid function, and poor general health are considered contraindications. Patients with rheumatoid arthritis and other systemic diseases can be expected to have poorer general health and vitality than those with uncomplicated degenerative joint disease. Poor general health may lessen the desirability of shoulder reconstruction even if the joint involvement is severe. Poor tissue quality, cuff deficiency, tuberosity, nonunion or malunion, remote infection, previous shoulder surgery, previous trauma, smoking, narcotic use, significant Parkinsonism, neuropathic arthropathy, obesity, crutch dependency, and unrealistic expectations all lessen the chances of a good result. Poor mental or emotional health may need management before shoulder reconstruction is undertaken; again, the routine preoperative use of the SF 36 may provide the surgeon with a "heads up" that these conditions exist. - The surgeon is experienced and prepared to provide a technically excellent arthroplasty.
In that reconstruction of the shoulder is fully as complex as that of the hip or knee, a similar type of learning and number of cases are necessary before mastery is achieved. According to the October 1993 to September 1994 National Inpatient Profile (HCIA INC 1995), the number of total knees performed in 12 months in the US was 211,872, while the number of total shoulders was only 5,895 (see footnote 1). These data indicate that surgeons have only 3% of the opportunity to master the total shoulder as they have to master the total knee. Expressed in another way, if the cases were distributed evenly, each of the 16,731 members of the AAOS would perform on the average just over one total knee per month and one total shoulder every three years.
The shoulder arthroplasty surgeon must have a command of the anatomy as well as the techniques to manage safely the exposure, capsular contractures, abnormalities of glenoid version cuff pathology, humeral deformities, and intraoperative problems. While the approximate number of cases to achieve mastery has not been determined, it is recognized that for shoulder, as well as for hip and knee, reconstruction "the surgeon is the method."
Goals of surgery
Shoulder arthroplasty provides the surgeon with the opportunity to restore the mechanics of glenohumeral motion, strength, stability, and smoothness.
- Motion is reestablished and obligate translation prevented by:
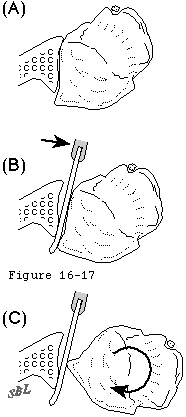
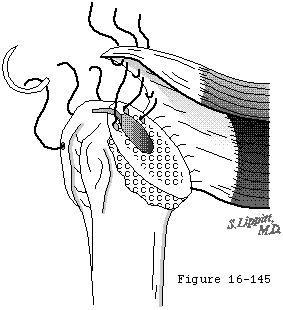
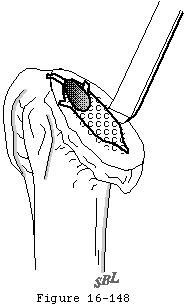
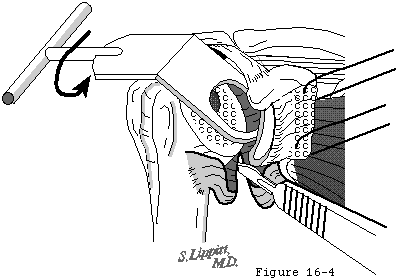
- releasing all adhesions and contractures at the humeroscapular motion interface (see figure 4).
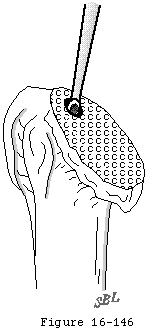
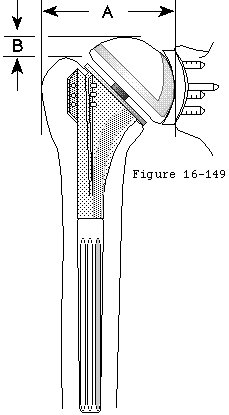
- inserting a smooth humeral prosthesis whose articular surface area comprises a substantial portion of the sphere (see figures 5 and 6).
- inserting a smooth glenoid prosthesis whose articular surface comprises a relatively small portion of the sphere (see figure 6).
- removal of blocking osteophytes (see figure 7).
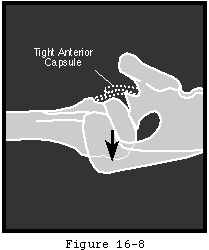
- avoiding overstuffing (see figure 8)
- Stability and Strength are achieved by:
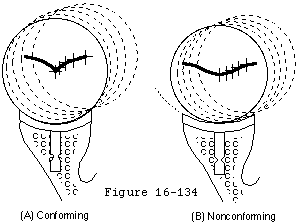
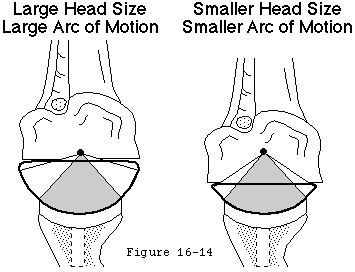
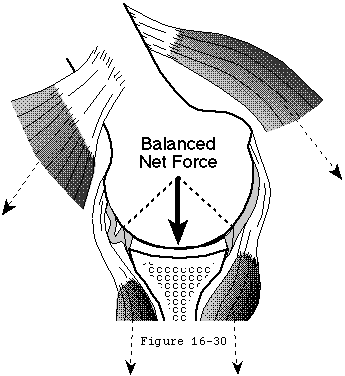
- normalizing glenoid and humeral joint surface location and orientation so that full surface contact occurs throughout the useful range of joint motion (see figure 9). Ballmer et al (Ballmer Lippitt Romeo et al. 1994) have studied in detail the effect of component articular surface geometry on the extent of glenohumeral joint surface contact. They found that for some commercially available prosthetic combinations, there was no position in which full surface contact existed, whereas others offer a 117 degree range of positions. They pointed out that in the range of positions where full surface contact exists there is no possibility for abutment of humeral bone or surrounding soft tissues against the glenoid edge. Furthermore, within this range, joint contact area is maximal, joint pressures are minimal, and the joint offers maximal stability. Conversely, outside the range of full surface contact, the edge of the glenoid may abut against humeral bone or soft tissue, there is increased joint contact pressure, and instability may result.
- selecting and positioning the new glenoid joint surface (see figures 10 and 11) so that effective arcs are available to balance the range of net humeral joint reaction forces usually encountered.
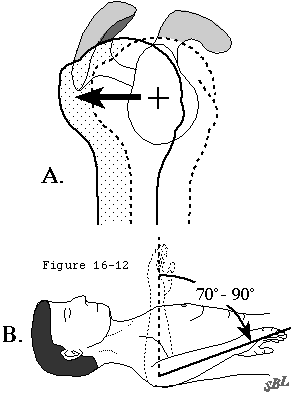
- reestablishing normal compressive muscle force (see figure 12) by releasing, repairing, balancing, and rehabilitating the cuff muscles (see figures 13 and 14)
The deltoid is the most important motor of the shoulder arthroplasty. The integrity of its origin, insertion, and nerve supply must be maintained. This is most easily accomplished by gently approaching the joint through the deltopectoral interval and by identifying and protecting the axillary nerve, both anterior-medially as it crosses the subscapularis and inferior capsule and laterally as it exits the quadrangular space and winds around the tuberosities on the deep surface of the deltoid. Rehabilitation of the deltoid is critical to the active motion following arthroplasty.
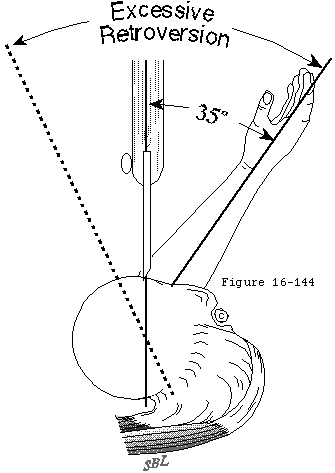
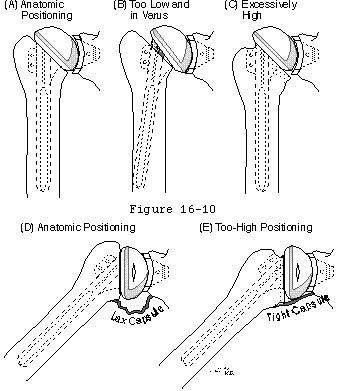
The rotator cuff mechanism is in jeopardy in shoulder arthroplasty for several reasons. The suprascapular nerve which supplies the supraspinatus and infraspinatus is at risk during surgical releases as it courses medial to the coracoid and then down the back of the glenoid 1 cm medial to the glenoid lip. The cuff tendons are at risk during surgery because the humeral cut must come close to their insertion to the tuberosities superiorly and posteriorly. A humeral cut made in excessive retroversion is likely to detach the cuff posteriorly and a cut made too low on the humerus is likely to detach the cuff superiorly (see figure 15). Overstuffing the joint places the cuff under tension when the arm is adducted or rotated (see figure 16). Most shoulder arthroplasties are performed for older individuals in whom the quality of the cuff tissue may be compromised, not only from age-related changes, but also from disuse enforced by chronic glenohumeral roughness. Shoulder arthroplasty may quickly restore motion and smoothness to the joint, placing new and substantial demands on the disused cuff tissue. Thus, the rehabilitation program and the patient's activities after arthroplasty must gradually increment the loads on the cuff allowing the tissue the opportunity to toughen over time.
If a cuff defect exists at the time of the arthroplasty, a cuff repair to bone should be carried out provided the quantity and quality of the cuff tissue are sufficient to allow a secure repair under physiologic tension with the arm at the side. If these conditions are not met, attempted cuff repair may not be worthwhile. If a cuff repair is carried out or if fixation of the tuberosities is performed, the rehabilitation after arthroplasty must be changed dramatically to allow for secure reattachment of the cuff mechanism to the humerus before active use is allowed.
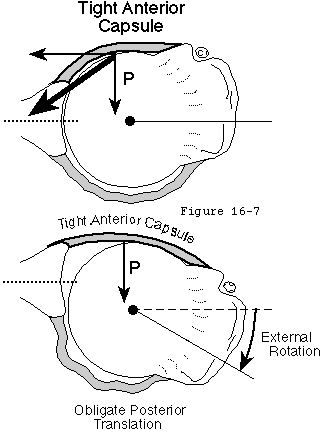
- assuring that capsular ligaments that are neither too short (in which case obligate translation may occur at the limits of motion) (see figure 17) nor too loose (in which case the joint may over-rotate beyond the positions in which the muscles can stabilize the head in the socket).
These considerations are critical. Recent reports (Rodosky and Bigliani 1994; Rodosky and Bigliani 1996; Rodosky Weinstein Pollock et al. 1995) suggest that nearly one-third of glenoid failures are associated with chronic glenohumeral instability after shoulder arthroplasty.
- normalizing glenoid and humeral joint surface location and orientation so that full surface contact occurs throughout the useful range of joint motion (see figure 9). Ballmer et al (Ballmer Lippitt Romeo et al. 1994) have studied in detail the effect of component articular surface geometry on the extent of glenohumeral joint surface contact. They found that for some commercially available prosthetic combinations, there was no position in which full surface contact existed, whereas others offer a 117 degree range of positions. They pointed out that in the range of positions where full surface contact exists there is no possibility for abutment of humeral bone or surrounding soft tissues against the glenoid edge. Furthermore, within this range, joint contact area is maximal, joint pressures are minimal, and the joint offers maximal stability. Conversely, outside the range of full surface contact, the edge of the glenoid may abut against humeral bone or soft tissue, there is increased joint contact pressure, and instability may result.
- Smoothness is provided by:
- inserting smooth prosthetic joint surfaces
- managing all humeroscapular motion interface roughness
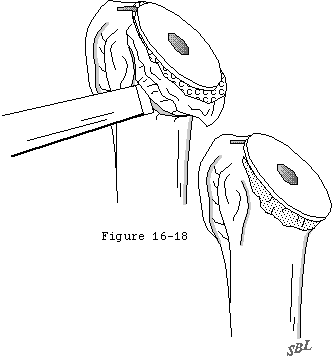
- implementing immediate postoperative motion to prevent unwanted scar formation (see figure 18). Early postoperative motion not only reduces the likelihood of adhesion formation but also increases the strength of soft tissue repairs. (Frank 1996)
Types of arthroplasty
Three levels of glenohumeral arthroplasty are commonly used. The basic surgical approach, capsular balancing, and osteophyte removal are similar for all three:
Non prosthetic arthroplasty is considered when osteophytes and capsular contractures block motion and function in the presence of congruent glenohumeral contact and reasonable cartilaginous space on radiographs. This option is particularly desirable in a young individual who plans to place heavy demands on the shoulder.
Prosthetic humeral hemiarthroplasty is considered when:
- The humeral joint surface is rough but the cartilaginous surface of the glenoid is intact (see footnote 2) and there is sufficient glenoid arc to stabilize the humeral head (see footnote 3). In this situation there is an even greater need to match the normal anatomy than that which exists with total glenohumeral replacement (see footnote 4).
- There is insufficient bone to support a glenoid component (for example after severe medial erosion of the glenoid in rheumatoid arthritis) (see figure 19)
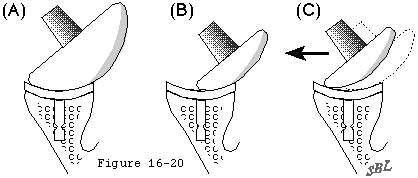
- There is fixed upwards displacement of the humeral head relative to the glenoid as in cuff tear arthropathy (see figure 20) or severe rheumatoid arthritis (see footnote 5).
- There is a history of remote joint infection and/or
- Heavy demands will be placed on the joint (as in motion disorders or anticipated heavy loading from occupation sport or lower extremity paresis).
Total glenohumeral arthroplasty is desirable when both joint surfaces are damaged and when both are reconstructable.
Recently, some studies have attempted to compare hemiarthroplasty and total shoulder arthroplasty. Boyd et al (Boyd Thomas Scott et al. 1990) found in a similar but unmatched series comparison that at 44 month followup hemiarthroplasty and total shoulder arthroplasty produced similar results in terms of functional improvement. Pain relief, range of motion, and patient satisfaction were better with total shoulder arthroplasty than hemiarthroplasty in the rheumatoid population. Progressive glenoid loosening was found in 12% of total shoulder arthroplasties, but no correlation with pain relief or range of motion was noted.
In a recent article, Rodosky and Bigliani (Rodosky and Bigliani 1996) reviewed the indications for glenoid resurfacing. They pointed out that early on in the history of shoulder arthroplasty, it was recognized that when the glenoid was significantly diseased, problems with excessive excursion of the prosthetic head were noted. The goals of inserting a glenoid prosthesis was to provide a better fulcrum and therefore better strength and greater stability (Neer 1985) along with decreased friction and elimination of "glenoid socket pain." (Rodosky and Bigliani 1996) Other potential benefits of the glenoid component include avoiding the progressive glenoid erosion seen when arthritis or fractures are treated with proximal humeral replacement alone. (Bigliani Flatow McCluskey et al. 1991; Compito Self and Bigliani 1994; Moeckel Dines Warren et al. 1992)
At this point the literature comparing hemiarthroplasty and total shoulder arthroplasty seems to favor the former when arthritis and cuff deficiency are coexistent (Arntz Jackins and Matsen 1991; Arntz Jackins and Matsen 1993; Brownlee and Cofield 1986; Codd Pollock and Flatow 1994; Cofield 1994; Fenlin Frieman and Allardyce 1995; Field Zubinski Dines et al. 1995; Kechele Basmania Wirth et al. 1995; Marmor 1977; Pollock Deliz McIlveen et al. 1992) and the latter in osteoarthritis and rheumatoid arthritis when the cuff is intact. (Bell and Gschwend 1986; Boyd Thomas Scott et al. 1990; Clayton Ferlic and Jeffers 1982; Cofield Frankle and Zuckerman 1993; Gschwend 1988; Neer 1985; Neer 1990; Neer Watson and Stanton 1982; Petersson 1986b; Pollock Higgis Codd et al. 1995) It is recognized that badly eroded glenoid bone cannot support a glenoid prosthesis. (Neer 1985; Neer 1990; Neer Watson and Stanton 1982)
Prosthesis selection
Desirable characteristics of the glenoid prosthesis
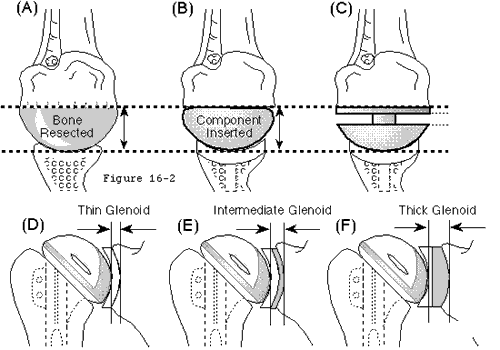
- As thin as structural properties will allow to minimize joint stuffing (see figure 8). For this reason all polyethylene components have an advantage because metal backing takes up needed room in the joint.
- Supported directly and intimately by bone (see figure 21 and figure 11) to avoid cracking away of a thin cement mantle (see footnote 6). The high incidence of failure of metal backed glenoid components has recently been recognized. (Rodosky and Bigliani 1994; Rodosky and Bigliani 1996; Rodosky Weinstein Pollock et al. 1995) Preservation of the subchondral bone and use of all polyethylene components results in loading patterns most similar to those found in a normal glenoid, whereas metal backed components lead to high non-physiologic stresses (Friedman LaBerge Dooley et al. 1992). Bone-prosthesis contact needs to be optimized by appropriate design, sizing, and bone preparation. Free-hand bone preparation is too uncertain to routinely provide optimal stability without resorting to the interposition of cement (see figure 21). Drill guides can assure that the fixation system achieves the desired relationship to the prepared glenoid face and minimizes the amount of bone removed.
- Fixation anterior and posterior to the meridian to prevent anterior and posterior rocking or "lift off" during eccentric loading (see figure 22).
- Fixation which preserves bone stock and minimizes the need for cement.
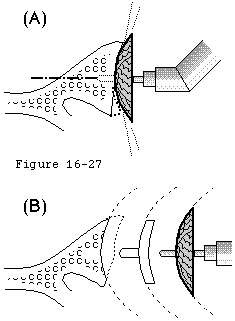
- Appropriate articular surface area (Ballmer Lippitt Romeo et al. 1994) and diameter of curvature relative to the humeral prosthesis (see figures 6, 23, 24, 25, and 26, and footnote 7.
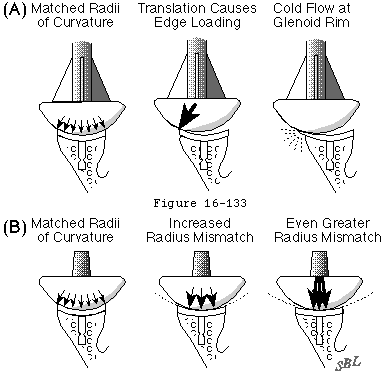
- Optimal articular surface diameter of curvature relative to that of the humerus. Harryman et al (Harryman Sidles Clark et al. 1990; Matsen Lippitt Sidles et al. 1994) demonstrated glenohumeral translation in normal shoulders with passive motion. Friedman et al (Friedman 1992) has used a radiographic technique to measure anteroposterior translation in 13 patients having Neer or Cofield total shoulder arthroplasties (in each of which the diameters of curvature of the glenoid and humeral surfaces were equal). They measured an average of 4 (range 0 to 12) mm of posterior translation between horizontal elevation in the -30° (posterior) plane and horizontal elevation in the 60° (anterior) plane. Along with Matsen et al (Matsen Lippitt Sidles et al. 1994) they pointed out that this translation could contribute to loosening or to asymmetric wear (see figures 23-25) as demonstrated by Collins et al. (Collins Tencer Sidles et al. 1992) Such a tendency for rim loading may be lessened if there is a slight diametrical mismatch between the humerus and the glenoid (see figures 23-26).
- Sufficient yield stress for the anticipated loading conditions (see figures 27 and 28).
- Normal orientation with respect to the scapula (see footnote 8).
Desirable characteristics of the humeral prosthesis
- Maximizes the percent of the sphere represented by the humeral articular surface area (see figures 4 and 5) (Ballmer Lippitt Romeo et al. 1994).
- Positions the humeral articular surface in the anatomic location and orientation.
- Provides secure humeral fixation in a way that preserves humeral bone stock (see footnote 9).
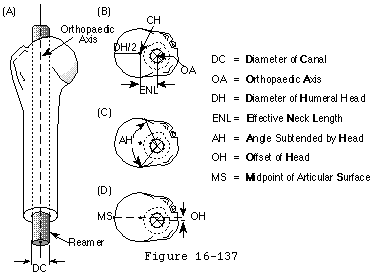
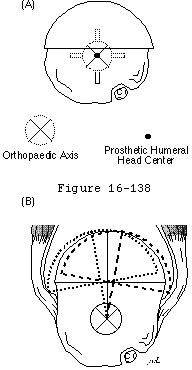
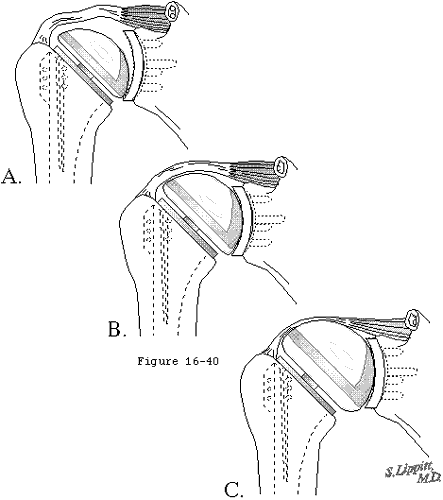
If the component is press fit in the medullary canal, the surgeon must recognize the restrictions this poses on the positioning of the prostheses. Ballmer et al (Ballmer Sidles Lippitt et al. 1993; Matsen Lippitt Sidles et al. 1994) pointed out that in a press fit situation, the canal, rather than the neck cut, becomes the primary determinant of the medial-lateral anteroposterior flexion-extension and varus-valgus position of the component. In fact with a snug canal fit, only two out of the six potential degrees of freedom remain: component height and component version. Canal fitting components usually are inserted after reaming the canal to the necessary depth and to a diameter judged safe and snug by the surgeon. The axis of this reamed proximal humeral canal is the "orthopedic axis" of the humerus (see figure 9). Again, the significance of this axis is that it defines much of the positional geometry of a humeral component press fit into it (see footnote 10).
Preferred surgical technique
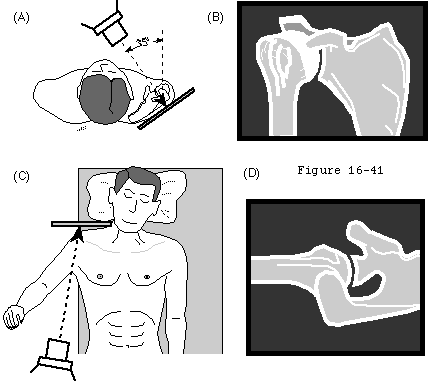
Standardized preoperative radiographs are obtained to reveal the amount quality and orientation of the glenoid bone as well as the size and configuration of the humerus down to where the tip of the humeral prosthesis will rest (see figures 2 and 3). Drawing the cuts and the implants on the preoperative radiographs using the manufacturer's templates helps the surgeon determine where the humeral and glenoid components should be positioned and whether any particular problems in their placement can be anticipated. Is there significant glenoid erosion or altered version? Are there potentially confusing glenoid osteophytes? Is there enough bone to support a glenoid component? What is the radius of the humeral joint surface? Is the humeral canal straight? What size is it? What is the position of the tuberosities in relation to the canal and the joint surface? How much humeral bone will need to be excised? Are there other major abnormalities of bony structure that could change the procedure? In press fit components will the medullary space accommodate the size and shape of the stem and the body of the prosthesis without risk of fracture?
After a brachial plexus block or general anesthetic, the patient is placed in the beach chair position with the thorax up at an angle of 30 degrees. The shoulder is just off the edge of the operating table so it can be moved freely through an entire range of motion. The anesthesiologist is positioned at the side of the neck on the opposite side from the shoulder being operated. A careful double skin preparation includes the entire arm and forequarter anteriorly and posteriorly. Draping allows access to the entire scapula clavicle and humerus.
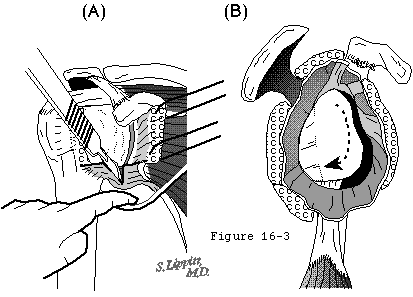
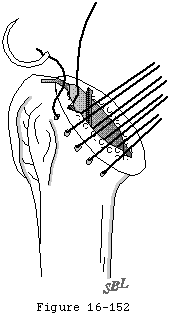
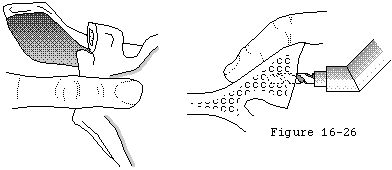
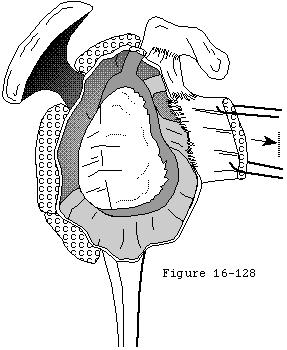
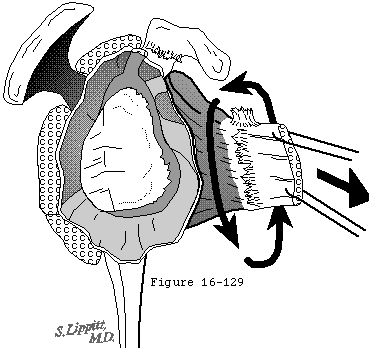
Skin incision is made over the deltopectoral groove along a line connecting the midpoint of the clavicle to the midpoint of the lateral humerus and crossing over the coracoid process (see figure 30). The deltopectoral interval is developed medial to the cephalic vein, preserving its major tributaries from the deltoid muscle (see figure 31). No deltoid detachment is needed proximally or distally. Incising the clavipectoral fascia at the lateral edge of the conjoined tendon up to but not through the coracoacromial ligament provides entry to the nonarticular humeroscapular motion interface (see figure 4). All adhesions in this interface are lysed from the axillary nerve medially to the axillary nerve as it exits the quadrilateral space posterior laterally. Burkhead et al (Burkhead Scheinberg and Box 1992) have recently provided an excellent review of the surgical anatomy of this nerve.
The subscapularis is incised at its insertion to the lesser tuberosity along with the subjacent capsule (see figure 32). This method of detachment maximizes the potential for a strong repair in that as Hinton et al (Hinton Parker Drez et al. 1994) point out the inferior 40% of the belly of the subscapularis extends all the way to the bone rather than inserting as a tendon. A 360 degree release of the subscapularis tendon is then performed assuring that it moves freely with respect to the glenoid the coracoid the coracoid muscles the axillary nerve and the inferior capsule (see figures 13 and 14).
Humeral preparation is the next step in the arthroplasty. The humeral head is exposed anteriorly by gentle external rotation and slight extension. Special care is exercised in old patients and in those with rheumatoid arthritis or other causes of fragile bone. Barriers to gentle external rotation may be unreleased anterior capsule or posterior osteophytes (see figure 33).
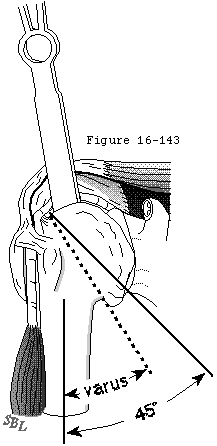
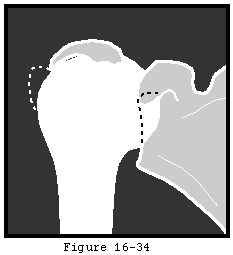
The humeral osteotomy requires attention to detail. While the degree of retroversion is often approximately thirty-five degrees, it may vary from ten to fifty. The ideal humeral cut is that which will allow positioning of the humeral prosthetic articular surface in the anatomic position. The cut plane must pass just inside the rotator cuff insertion to the tuberosity resecting the humeral articular surface without damaging the cuff insertion (see figures 34 and 35). In degenerative joint disease, the apparent articular surface may not provide an accurate indication of the plane of humeral head resection. The angle of the cut with the humeral shaft must match that of the prosthesis being used - often about 45 degrees. The amount of humeral bone to be resected is compared for the different prosthetic options (see figure 8).
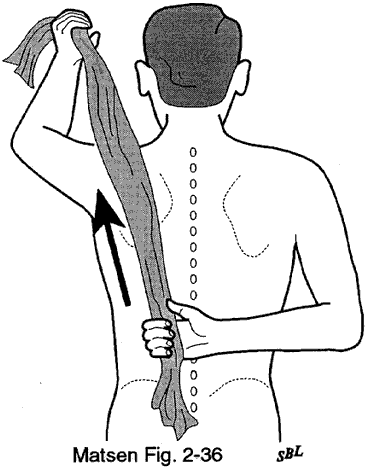
After the humeral osteotomy the surgeon can get an idea of the joint volume remaining for the glenoid and humeral head components by pushing the humeral neck laterally with a finger. This step is helpful in determining the need for further soft tissue releases. If the capsule is so tight that even the smallest head will not fit, more release is required (see figure 36). Cutting away more humerus is not an option because the humeral head has already been resected at the cuff insertion and further resection will jeopardize this essential attachment.
With the proximal humerus displaced medially into the joint, the rotator cuff is palpated to establish its integrity. If a repairable defect through quality cuff tissue is identified, the retracted tendon is mobilized so it will reach the tuberosity without undue tension with the arthroplasty components in place and with the arm at the side (see figure 37).
However, the two potential downsides of cuff repair in this circumstance are recognized:
- In the presence of deficient tendon, cuff repair tightens the glenohumeral joint and
- Cuff repair changes the post op rehabilitation from active to passive motion until the tendon has healed.
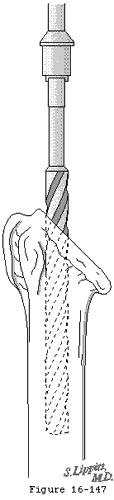
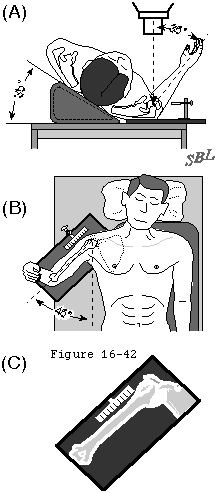
The medullary canal of the humerus is reamed starting at a point lateral on the cut surface just behind the bicipital groove (see figure 38). Starting with a small diameter reamer, reaming is continued up to the diameter appropriate to the component using a slight valgus bias and while protecting the biceps and cuff (see figure 39). For prostheses having press fit stems, medullary reaming continues until a snug fit is achieved. This press fit limits the degrees of freedom for placing the humeral component (see figure 40). If necessary, slots are made in the tuberosity to accommodate the fins and throat of the component (see figure 41). The slot for the lateral fin should be just posterior to the bicipital groove. A trial component body is inserted so that the prosthetic neck is centered on the neck of the bony humerus. The trial component is used as a guide to the excision of the osteophytes all around the humeral neck (see figure 7). Ideally the horizontal and vertical distances between the tuberosity and the joint surface should be normalized (see figure 42).
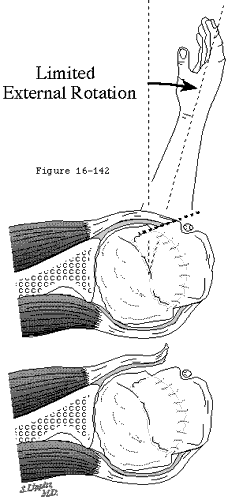
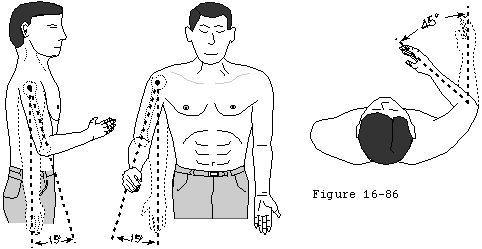
By placing various trial humeral heads the surgeon can select the size that allows 70 degrees of internal rotation of the abducted arm (the "scarecrow" test) and 15 mm of translation on the posterior drawer test (see figure 43). These two parameters are guides to the posterior capsular laxity usually necessary to achieve a satisfactory range of motion. A global periglenoid capsular release may be necessary to achieve this laxity (see figure 36 and figure 44). However, in degenerative joint disease pre-operative posterior subluxation usually obviates the need for posterior capsular release. The important interplay between humeral component position and capsular laxity is recognized (see figure 45).
Preparation of the glenoid
Accurate preparation of the glenoid bone requires the excellent surgical exposure that results from humeral head and osteophyte excision and appropriate capsular release.
The goals of the glenoid part of the arthroplasty are;
- Normalized glenoid orientation
- Direct support of the component by precisely contoured bone
- Secure fixation and
- Avoidance of overstuffing (see figure 8).
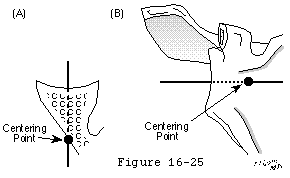
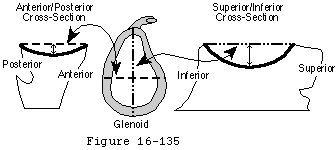
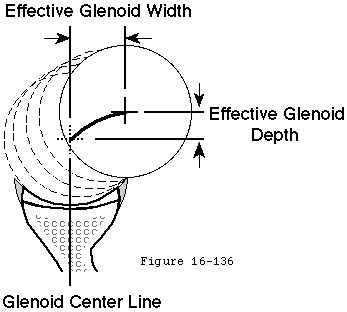
Glenoid orientation is defined in terms of the glenoid center line: the line perpendicular to the center of the normally oriented glenoid face. The shoulder arthroplasty surgeon should practice verifying the landmarks for a normal glenoid center line by drilling holes perpendicular to the glenoid face of normal cadaveric scapulae and observing their exit in a consistent spot just medial to the anterior scapular neck: the "centering point" (see figure 46). This spot lies between the upper and lower crus of the body of the scapula as they approach the neck. After the capsular releases have been performed at surgery, this centering point can be palpated at the lateral extent of the subscapularis fossa. Because the location of this centering point is unaffected by arthritis, it is of great value in normalizing the orientation of a distorted glenoid face. It is particularly useful in correcting the posterior facing of the glenoid face that commonly results from posterior erosion in degenerative joint disease.
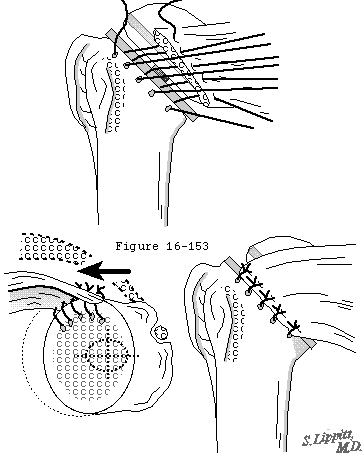
An index finger identifies the centering point on the anterior scapular neck while a hole is drilled from the center of the glenoid face toward it (see figure 10). The orientation of the glenoid face is normalized using a spherical reamer with a guiding peg inserted along the glenoid center line drill hole (see figure 11). Appropriate positioning of retractors facilitates this reaming (see figure 47). This technique is usually sufficient to manage posterior erosion; posterior glenoid bone grafting (see figure 48) is rarely necessary. If there are reasons not to insert a glenoid component, this normalizing reaming provides an excellent non-prosthetic glenoidplasty.
Once the reaming is completed, the glenoid center line hole and the reamed glenoid surface can be used to orient precisely a drill guide for making additional fixation holes as required by the particular glenoid component design. Each hole is checked to determine whether it penetrates the scapula at its depth. Penetrating holes are cemented but the cement is not pressurized.
A glenoid component is selected that covers the maximal amount of the prepared glenoid face with minimal overhang. The quality of the glenoid bone preparation is checked by inserting the glenoid trial and ensuring that it does not rock even when the surgeon's finger applies an eccentric load to the rim.
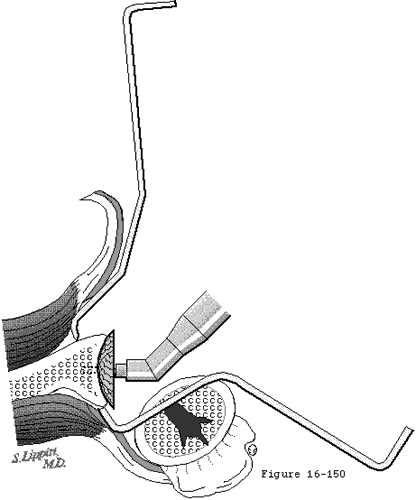
After water spray irrigation, the holes are cleaned and dried with a spray of sterile CO2 gas (Innovative Surgical Devices Stillwater MN 55082). A small amount of cement is injected into each of the holes with a large-tipped syringe. Holes that do not penetrate the scapula can be pressurized by the syringe. No cement is placed on the bony face of the glenoid; if the back of the glenoid component matches the prepared bony face, there is no advantage of an interposed layer of cement which could fail and displace leaving the glenoid component relatively unsupported. Contact between precisely contoured bone and polyethylene ("bone backing" as opposed to metal backing) provides an optimal load transfer mechanism (see figure 21). After the glenoid component is pressed into position the absence of residual cement bits in the posterior shoulder is verified.
Insertion of the humeral body is the next step in the arthroplasty. Prior to the insertion of the body the surgeon places at least six sutures of #2 nonabsorbable suture in secure bone at the anterior humeral neck for later attachment of the subscapularis tendon (see figure 49). Final balancing of the soft tissues must be verified before the definitive humeral component is inserted.
A shoulder arthroplasty with balanced soft tissues should allow:
- 70 degrees of internal rotation of the arm elevated in the coronal plane ("scarecrow" test)
- 15 mm of posterior subluxation of the humeral head on the posterior drawer test
- 140 degrees of elevation and
- 40 degrees of external rotation of the unelevated arm with the subscapularis approximated (see figure 43). A tighter shoulder will not only have limited range of motion but may also challenge the rotator cuff (see figure 16) and foster obligate translation at the extremes of motion with resultant rim loading risking glenoid loosening and component deformation (see figure 22). Some component systems are designed to allow a small amount of translation before rim loading occurs (see figures 23-25).
The humeral component is then inserted into the prepared proximal humerus. The height version and fixation are carefully checked. If sufficient stability of the prosthesis in the bone is not achieved with a press fit bone graft or cement may be used.
Prior to closure the wound is thoroughly inspected for debris. The joint is put through a full range of motion to verify smoothness and lack of unwanted contact for example between the medial humerus and inferior glenoid ("Pooh Corner"). The wound is drained. The subscapularis is repaired securely to the humeral neck so that the unelevated arm can be externally rotated by 40 degrees (see figure 50). If additional subscapularis length is required a "Z-plasty" can be performed although this compromises the strength of the tendon. The wound is closed in layers. Simple interrupted skin sutures are preferred when substantial drainage is anticipated or when wound healing may be impaired (such as in an individual on corticosteroids or with thin rheumatoid skin).
Footnotes
Footnote 1: Similar data are presented by Madhok et al (Madhok Lewallen and Wallrichs 1993) who reviewed the trends in utilization in upper limb replacements at the Mayo Clinic from 1972-90.
Footnote 2: The articular cartilage is assessed by preoperative radiographs and at surgery by observation palpation and by listening to the sound when it is struck with a small blunt elevator: thin cartilage or bare bone will cause the elevator to ring while normal cartilage will yield only a dull "thunk."
Footnote 3: Frequently in degenerative joint disease and in capsulorrhaphy arthropathy the posterior half of the glenoid concavity is eroded away depriving the shoulder of the effective glenoid arc (see figure 51). Thus even if excellent articular cartilage exists on the anterior half of the glenoid a humeral hemiarthroplasty cannot be stable without this posterior glenoid lip.
Footnote 4: When performing a hemiarthroplasty the goal is to restore the humeral articular surface to its normal location and configuration. Because the glenoid is not replaced the size radius and orientation of the prosthetic humeral joint surface must duplicate that of the original biological humeral head. If necessary the details of the patient's normal humeral head anatomy can be best obtained from radiographs of the opposite shoulder.
Footnote 5: Humeral hemiarthroplasty may be stabilized in cuff tear arthropathy even though the superior lip of the glenoid is eroded away by superior humeral subluxation. In this situation the prosthetic humeral head is captured by an acetabular-like socket consisting of the eroded upper glenoid the coracoid the coracoacromial ligament and the acromion provided these structures have not been sacrificed by acromioplasty. Again it is vital that the surgeon not compromise this socket by sacrificing the anterior acromion or the coracoacromial ligament; otherwise the humeral head is likely to be destabilized in an anterosuperior direction.
In hemiarthroplasty for cuff tear arthropathy the undersurface of the "acetabularized" coracoacromial arch is usually polished smooth with a consistent radius of curvature. The prosthetic humeral articular surface and the tuberosities must provide a smooth congruent surface to mate with this arch. Achieving this goal requires attention to the selection and positioning of the humeral component and to sculpting the tuberosities. The best choice is a humeral prosthesis which duplicates the size and position of the humeral head which is excised. The large smooth joint contact area achieved in this procedure appears to be responsible for its success in restoring comfort and function in the difficult problem of cuff tear arthropathy.
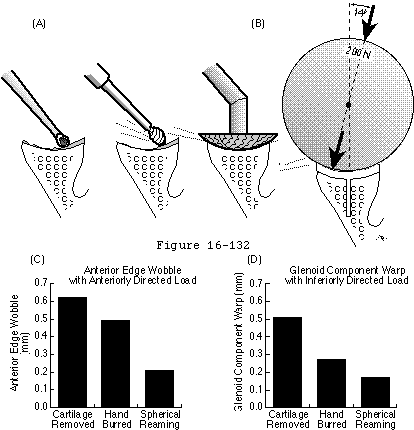
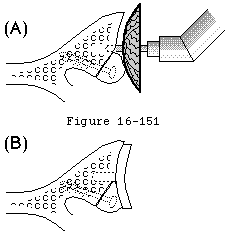
Footnote 6: In order for the glenoid to stabilize the humeral head against transverse loads it must be well supported by the bone beneath it. Clinical observations suggest that a primary mechanism of glenoid loosening is via the rocking horse mechanism when eccentric loads are applied. In a series of 10 cadaver glenoids the authors studied the effect of glenoid bone preparation on the stability of a 3 mm thick non clinical glenoid component with a diameter of curvature of 60 mm on the surface apposed to bone. (Matsen Lippitt Sidles et al. 1994) To emphasize the effect of glenoid surface preparation the component was secured to the bony glenoid with only a single flexible uncemented central peg. The component was loaded with an eccentric force of 200 Newtons applied at an angle of 14 degrees with the glenoid center line. While the component was loaded the wobble of the component with respect to the bone and the warp or deformation of the component were measured using displacement transducers.
The stability of the component was measured sequentially after three different glenoid preparations:
- Curettage of the articular cartilage
- Meticulous burring of the bone by hand to fit the back of the component and
- Preparation using a reamer with a diameter of curvature of 60 mm centered in a hole along the glenoid center line. Spherical reaming dramatically diminished both the wobble and the warp of the glenoid component with eccentric loading in comparison with the other two methods of bone preparation (see figure 21). It is likely that an even greater increment in stability would accrue with concentric reaming in a deformed bony glenoid such as that found in degenerative joint disease. This study demonstrates that precise contouring of the bone to fit the back of the glenoid component provides excellent support of the prosthesis even without the potential benefits of fixation using multiple pegs, keels, cement, screws, or tissue ingrowth.
Spherical reaming along the anatomic glenoid center line has two important advantages:
- It normalizes glenoid version and
- It provides "bone back" support of the glenoid component with the opportunity for optimal stability and load transfer without the use of metal-backing.
Footnote 7: Glenohumeral arthroplasty provides the surgeon the opportunity to control the shape of the prosthetic glenoid concavity. The depth of the glenoid concavity is related to dimensions of the face of the glenoid (superior-inferior and anterior-posterior breadth) and to the radius of curvature set. For a given radius of joint surface curvature larger components are deeper than smaller ones. For a given glenoid size components with a smaller radius of curvature are deeper than those with larger radii of joint surface curvature.
If the glenoid and humeral radii of curvature are equal, the head will be held precisely in the center by concavity compression; no translation can occur unless the humeral head is allowed to lift out of the fossa (see figure 53). While this tight conformity provides excellent stability it has the potential disadvantage that displacing loads applied to the humerus will be transmitted fully to the glenoid and thence to the glenoid-bone interface. In the biological glenoid the compliance of the articular cartilage and glenoid labrum provide shock absorption for these transverse displacing loads. Because polyethylene is much stiffer than cartilage and labrum this shock absorption is not present in prosthetic glenoid arthroplasty. Thus glenoid fixation is at risk for substantial peak loads when the glenoid and humeral joint surfaces are totally conforming.
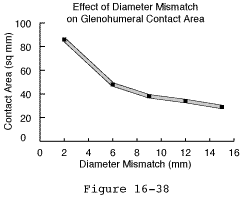
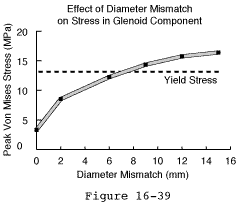
Some degree of shock absorption can be provided by a slight mismatch between the humeral and glenoid radii of curvature that of the glenoid being slightly larger. This allows some translation before the humeral head must lift out of the fossa (the glenoidoram becomes more a "U" than a tight "V") (see figure 26). This too is a compromise however in that the degree of mismatch decreases the contact area and increases the contact pressures with potential risk of polyethylene failure. In a finite element model using conventional polyethylene the surface area of contact with a load of typical body weight 625 Newtons (140 lb) was predicted to decrease dramatically with increasing degrees of radial mismatch (see figure 40). This drop in contact area gives rise to a corresponding increase in the contact stresses (see figure 28) For loads of 625 Newtons the contact stress exceeds the predicted yield stress for conventional polyethylene when the radial mismatch is greater than 3.0 mm.
Footnote 8: A simple cadaver study demonstrated a practical method for normalizing the glenoid orientation. The center of the face of the glenoid was located in each of ten normal cadaveric scapulae. A drill was then inserted perpendicular to the face starting at the glenoid center. In each case the drill emerged from the anterior glenoid neck at the lateral aspect of the subscapularis fossa at a point midway between the upper and lower crus of the scapula (see figure 46). This spot is known as the "centering point." This point is easily palpated at arthroplasty surgery after an anterior capsular release has been performed (see figure 10). The line connecting it to the center of the glenoid face is the normalized glenoid center line. Orienting the prosthetic glenoid to this normalized glenoid center line enables the surgeon to correct pathologic glenoid version which is frequently encountered in degenerative joint disease and other conditions requiring shoulder arthroplasty (see figure 11).
Footnote 9: Good fit and fill of the humerus can often provide secure fixation without cement but press fitting does increase the risk of humeral fracture. Whether the medullary canal needs to be sealed to prevent entry of polyethylene debris remains a theoretical consideration.
Footnote 10: Using this axis as a reference several geometric parameters were measured in ten cadaveric humeri ranging in age from 37 to 78 years (mean 60 years). The results are shown in the Table 16-19.
For components that fit snugly within the medullary canal, changes in humeral version must take place about the orthopedic axis. This does not allow much latitude in the version if the humeral articular surface is to be optimized (see figure 40). For this reason and because the center of rotation of the head lies close to the orthopedic axis, the soft tissue tension is not substantially changed by alterations in humeral version (a very different situation from that encountered in the hip where the center of rotation of the head is distant from the medullary axis of the shaft). Ballmer et al (Ballmer Sidles Lippitt et al. 1993) found that only a 2 millimeter change in combined head/neck length could be achieved by change in version of a prosthesis press fit in the canal.
Zuckerman (Zuckerman 1996) compared hospital reimbursement by Medicare for DRG 491 (shoulder arthroplasty) and found an overall range from $4699 to $9856 with a mean for urban locations of $6906 and for rural location of $5198. He found the cost of humeral components ranged from $950 to $2250 while the glenoid components ranged from $520 to $1250. Total shoulder systems ranged from $1470 to $2900. Thus if the least costly implant system was used in a location with the greatest DRG reimbursement it would account for $1470/$9856 or 15% of the hospital reimbursement. At the opposite extreme if the most costly implant was used where DRG reimbursement was the least it would account for $2900/$4699 or 62% of the total hospital reimbursement.
Special considerations in arthroplasty
Degenerative joint disease
In this condition the glenoid face is typically flattened and often eroded posteriorly from chronic posterior subluxation (see figure 54). The glenoid may be distorted by peripheral osteophytes masking the location of the anatomic fossa. The humeral head may be flattened in a corresponding manner and effectively enlarged by the proliferation of "goat's beard" osteophytes from the anterior inferior and posterior articular rim. Intraarticular loose bodies may lie hidden in the subcoracoid or axillary recesses. Anterior capsular and subscapularis contractures are common in degenerative joint disease and require release. However, posterior capsular release is not performed if there is posterior humeral subluxation preoperatively.
Rheumatoid arthritis
The basic principles of shoulder arthroplasty in rheumatoid arthritis are similar to those in degenerative arthritis but some important differences exist. Rheumatoid tissues are much more fragile. The bone is more prone to fracture and the muscle and tendons are more prone to tear. Thus, from the outset, extreme care must be taken to preserve bone and soft tissue integrity. We refer to these requirements for extraordinary gentleness as "rheumatoid rules."
Because rheumatoid arthritis is an erosive and destructive disease, tissue deficiencies of the bone and rotator cuff are more likely than in degenerative joint disease. Thus, the soft tissues anteriorly may be insufficient to allow for a subscapularis lengthening. The glenoid bone may be so eroded that there is insufficient stock to support a glenoid component. The rotator cuff may be partially or totally deficient. Thus, in the preoperative evaluation and in discussion with the patient concerning the possible outcomes of surgery all of these factors need to be considered.
The standard preoperative scapular anteroposterior and axillary radiographs are required to evaluate the humeral and glenoid bone stock. In rheumatoid arthritis the glenoid erosion is usually medial (rather than posterior as in degenerative joint disease). For this reason only minimal glenoid reaming may be necessary to achieve an excellent quality fit to the back of the glenoid component. The potential fragility of the bone and soft tissues makes it particularly important that the joint not be overstuffed and that adequate soft tissue laxity be present for immediate postoperative motion. This is particularly a challenge in diminutive patients with juvenile rheumatoid arthritis who may also have a tiny humeral medullary canal. In some cases there may be insufficient joint volume to permit the insertion of a glenoid component in spite of complete soft tissue releases.
Secondary degenerative joint disease
In posttraumatic arthritis the challenges may be even greater. The anatomy is likely to be distorted by previous fracture and surgery. The nonarticular humeroscapular motion interface is likely to be scarred obscuring important neurologic structures such as the axillary nerve. The tuberosities the humeral shaft and the glenoid may be ununited or malunited.
As a first step the motion interface must be carefully freed and the axillary nerve identified both as it crosses the subscapularis and as it courses laterally on the deep surface of the deltoid. Case by case judgments must be made concerning the need for osteotomy to try to restore more normal anatomic relationships recognizing that additional healing and postoperative protection may be required. Again the goal is restoration of anatomic relationships firm fixation of components soft tissue balance stability and smooth gliding in the humeroscapular motion interface.
Capsulorrhaphy arthropathy
Shoulders affected by capsulorrhaphy arthropathy present additional challenges such as neurovascular scarring from previous surgery, soft tissue contractures, bone deficiencies, implants from previous surgery, changes of glenoid version, and an increased potential for glenohumeral instability after the arthroplasty (see figure 55).
Cuff tear arthropathy
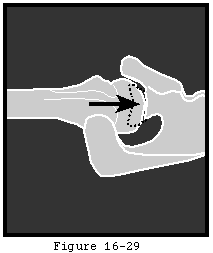
In this condition there are several unique challenges for regaining glenohumeral smoothness. The humeral head is subluxated in a superior position so that it is articulating with the coracoacromial arch. The rotator cuff is almost never amenable to a strong repair and the glenoid is eroded superiorly so that an acetabular-like structure is formed in continuity with the coracoacromial arch. Under these circumstances normal glenohumeral relationships are very difficult to normalize and maintain by a durable cuff reconstruction. More often it is preferable to accept the altered joint relationship which uses the "acetabulum" for secondary stability in the absence of primary stability from the rotator cuff. In this "special hemiarthroplasty the articular surface of the proximal humerus is resurfaced with a component matching the preoperative humeral joint surface size and position. The tuberosities are smoothed so that they are congruous with the humeral articular surface. This allows for the proximal humerus to match the acetabulum and to articulate smoothly within it. It is very important to avoid using oversized" humeral components because they overstuff the joint, do not match the concavity of the "acetabulum," and restrict joint motion. In a special hemiarthroplasty the patient is spared the necessity of protecting a rotator cuff repair so that immediate passive and active exercises can be instituted after surgery. The patient is also spared the risk of glenoid loosening from the rocking horse mechanism (see figure 56).
The ideal patient for this procedure has a normal deltoid muscle, a concentric coracoacromial "acetabulum" stabilizing the proximal humerus which is superiorly displaced with respect to the glenoid, concentric erosion of the upper glenoid fossa, a "femoralized" upper humerus with rounding off of the greater tuberosity, an irreparable rotator cuff defect, no previous surgical compromise of the acromion or coracoacromial ligament, good patient motivation, and realistic expectations.
In a series of ten patients having special hemiarthroplasty for rotator cuff tear arthropathy, the range of active motion and function were substantially improved by this procedure (Table 16-20). These results are not be as good as those for total glenohumeral arthroplasty because the patient lacks the benefit of both prosthetic glenoid smoothness as well as the function of the rotator cuff.
Postoperative rehabilitation
Continuous passive motion
Rehabilitation is started immediately following surgery in the recovery room with the initiation of slow and gentle continuous passive motion (see figure 57).
For patients who have had an interscalene block prior to surgery, this early motion is pain free. Continuous passive motion (CPM) should be stopped and the wrist brace removed every two to three hours for approximately 15 - 20 minutes to relieve any skin and nerve compression. A sling is worn between exercise sessions until active muscle control is regained.
The patient-conducted rehabilitation program is started the day of surgery under the instruction of the surgeon or therapist. While the program may vary with the details of the surgery performed (see footnote 1) the following is a description of the basic program for shoulder arthroplasty. The stretching exercises include
- elevation
- external rotation limited to 40 degrees
- internal rotation
- cross body adduction
- grip strengthening
- elbow range of motion
- external rotator isometrics and
- anterior middle and posterior deltoid isometrics.
The patient is instructed to perform a total of five exercise sessions spread evenly throughout the day both while in the hospital as well as at home following discharge.
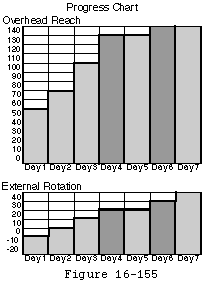
Charts are placed on the wall in full view from the patient's bed to graph the progress of external rotation and elevation (see figure 58) as measured by the surgeon or therapist. This provides positive feedback for rehabilitative progress.
For the routine arthroplasty the range of motion goals to be achieved before discharge are 140 degrees of elevation 40 degrees of external rotation and functional internal rotation and cross body adduction. These goals may be modified according to the specific surgical procedure. Because the desired range has been achieved on the operating table the patients' task is simplified they have only to maintain this range during the post operative period.
Exercises
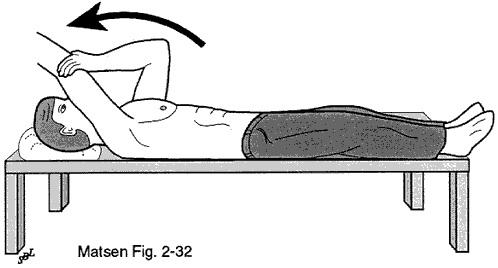
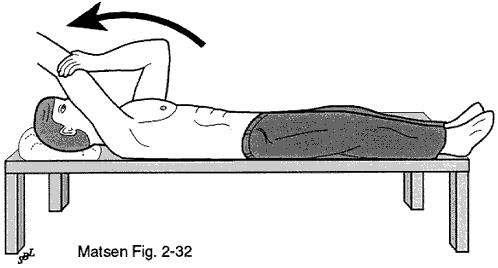
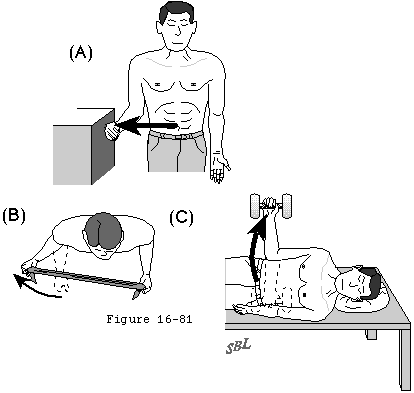
Elevation (overhead reach) is performed in the supine position (lying flat on the back) grasping the wrist or elbow of the operative shoulder with the hand of the unoperated arm pulling up toward the ceiling and reaching overhead as high as possible to the goal of 140 degrees with the arm relaxed (see figure 59). A pulley or the forward lean (see figure 60) may also be useful in achieving elevation especially if the opposite shoulder is involved.
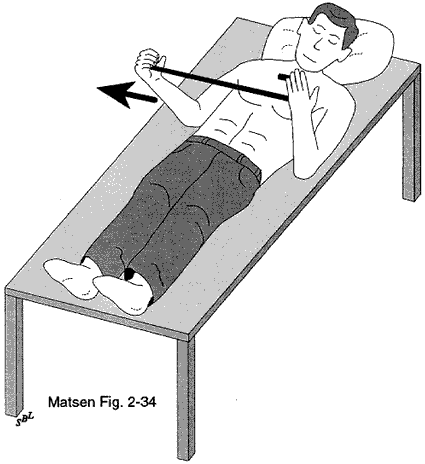
External Rotation (rotation away from the body) is performed in the supine position (see figure 61) the operative side elbow held against or close to the side and flexed to 90 degrees. A stick is held in both hands so that the unaffected extremity pushes on the operative arm to externally rotate it to the goal of 40 degrees. Holding on to a door and turning away is another useful way to stretch external rotation (see figure 62).
Internal Rotation is performed by grasping the wrist of the relaxed involved arm with the non-operative side hand the hands are lifted up the back as high as possible. A towel can also be used to assist with pulling the involved arm into internal rotation behind the back (see figure 63).
Cross Body Adduction is performed sitting or standing grasping the elbow of the involved arm with the other hand. The involved arm is relaxed with the elbow extended and pulled across the body until a stretch is felt (see figure 64).
Active Elbow Motion is performed standing in order to allow unimpeded or unrestricted flexion/extension and supination/pronation.
Grip Strengthening is performed to maintain forearm tone and can be accomplished using a foam pad or tennis ball.
External Rotator Isometrics are performed with the forearm at neutral rotation. An attempt is made to move the wrist out to the side against the resistance of the other hand or a fixed object (see figure 65).
Deltoid Isometrics are also performed standing or sitting. The arm is held in a neutral position and pushed forward to the side and to the back to exercise the anterior middle and posterior deltoid respectively.
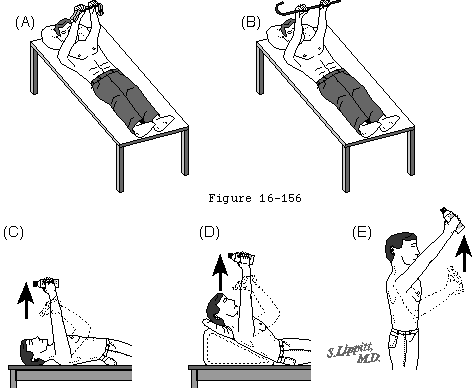
Supine Presses are performed initially holding a cloth or stick between both hands with the hands held close together (see figure 66). From a starting position with the elbows bent and hands lying across the chest the stick is pushed straight to the ceiling with both hands in a slow and controlled manner and then slowly lowered back to the resting position at the chest. The space between the two hands is progressively increased. As the shoulder becomes stronger the hands are pushed to the ceiling in a slow and controlled manner independent of each other. With increasing strength the exercise is conducted with a one pound weight which is held in the involved hand as it is pressed to the ceiling. When that is comfortable the incline is gradually increased to eventually reach the upright position. All presses should be performed in a slow and controlled manner; they are progressed to the next level only when 20 repetitions can be performed comfortably.
The patient is instructed in all these exercises on three occasions:
- Prior to surgery
- Immediately after surgery and
- Prior to leaving the hospital. Before discharge the goals of assisted external rotation to 40 degrees and assisted elevation to 140 degrees must be accomplished.
The patient is placed in charge of their own rehabilitation and taught to progressively return to normal use of the shoulder. Typically keyboarding and driving are achieved at two weeks, swimming is started at six weeks, golf or tennis are started at three-six months, and chopping wood is precluded.
Footnotes
Footnote 1: For example, if tuberosity or cuff fixation has been part of the procedure, external rotation isometrics and active elevation may be delayed. Ernest A. Codman will be remembered in the annals of orthopedic history as a pioneer in the study of shoulder disorders but few except the most ardent of his followers are familiar with the role he played as a visionary and champion of what is currently described as outcomes research. (Codman 1934)
Methods of assessing functional outcome
Ernest A. Codman will be remembered in the annals of orthopedic history as a pioneer in the study of shoulder disorders, but few except the most ardent of his followers are familiar with the role he played as a visionary and champion of what is currently described as outcomes research. (Codman 1934)
Evaluating success and failure
Central to what Codman described in the early 1900's as an "end-result" system was the admonition that every patient should be followed to determine if the treatment was a success and if not to determine the reasons for failure so that such occurrences could be prevented in the future. Despite Codman's admonition nearly 100 years ago there continues to exist a lack of standardized methods for measuring results and reporting complications associated with total shoulder implants. Unfortunately the lack of a universally accepted outcome measurement system for shoulder arthroplasty increases methodologic flaws in structured literature reviews and often precludes meaningful retrospective or prospective comparisons between various arthroplasty series. The desirability of research methodologies which will improve the quality and comparability of multicenter studies is underscored by our review of nearly fifty total shoulder replacement series. (Boyd Thomas Scott et al. 1990; Brenner Ferlic Clayton et al. 1989; Brostrom Kronberg and Wallensten 1992; Brumfield Schilz and Flinders 1981; Clayton Ferlic and Jeffers 1982; Cofield and Daly 1992; Cofield and Stauffer 1977; Coughlin Morris and West 1979; Cruess 1980; Engelbrecht Siegel Rottger et al. 1980; Faludi and Weiland 1983; Fenlin 1975; Fig.gie Inglis Fig.gie et al. 1989; Frich Moller and Sneppen 1988; Gristina Romano Kammire et al. 1987; Gristina and Webb 1982; Hawkins Bell and Jallay 1989; Kelly Foster and Fischer 1987; Kolbel Rohlmann and Bergmann 1982; Laurence 1991; Lettin Copeland and Scales 1982; Linscheid and Cofield 1976; Martin Sledge Thomas et al. 1995; Mazas and de la Caffiniere 1981; McCoy Warren Bade et al. 1989; McElwain and English 1987; McKeeand Watson-Farrar 1966; Neer and Morrison 1988; Neer and Kirby 1982; Pahle and Kvarnes 1985a; Pollock Deliz McIlveen et al. 1992; Post 1987; Post Haskell and Jablon 1980; Post and Jablon 1983; Post Jablon Miller et al. 1979; Roper Paterson and Day 1990; Torchia Cofield and Settergren 1994-1995; Warren Ranawat and Inglis 1982; Wilde Borden and Brems 1984) The results of this review revealed that only thirty-three of these reports assessed the outcome of treatment by applying a specific grading system. Moreover there was a great lack of unanimity in regards to these evaluation schemes as twenty-two different grading systems were utilized. Also disconcerting was the variability in reported data variations in terminology and ill-defined standards of assessing complications which made it difficult to systematically analyze many of these studies.
The necessity for improving study design defining the important constituents of outcome measurement and increasing the validity of orthopedic clinical research has been emphasized by several authors. (Cowell and Curtiss 1985; Cutler and Ederer 1958; Gartland 1988; Rudicel and Esdiale 1985) It has been suggested that the current emphasis of orthopedic clinical studies should be directed toward outcome research which documents the effect of treatment on the health of those treated and the subsequent quality of their lives. (Gartland 1988) Recently the American Shoulder and Elbow Surgeons proposed a standardized form for assessment of the shoulder which is applicable to all patients regardless of diagnosis. (Richards An Bigliani et al. 1994) Such standardized forms represent assessment tools which will facilitate the analysis of multi-center studies permit validity testing of measurement tools and provide documentation of patient outcome in terms of economics and improved quality of life.
Survivorship analysis
The validity of survivorship analysis in the evaluation of long-term clinical studies involving total hip replacements is well established. (Cornell and Ranawat 1986; Dobbs 1980; Dorey and Amstutz 1986; Jinnah Amstutz Tooke et al. 1986; Pavlov 1987) Although non-parametric estimates of survivorship based upon life tables and the Kaplan-Meier curve have proven useful in predicting the longevity of hip arthroplasties the application of these instruments to total shoulder arthroplasty studies is limited to two series. (Brenner Ferlic Clayton et al. 1989; Cofield 1993)
In 1983 Cofield performed a non-parametric estimation of survivorship in 176 unconstrained total shoulder arthroplasties and predicated a 9.6 percent cumulative probability of failure at five years. (Cofield 1993) The criteria for failure was defined as the need for a major reoperation which occurred in eight (4.5 percent) cases. The indication for reoperation included early dislocation in three shoulders glenoid component loosening in three shoulders and muscle transfer for axillary nerve paralysis and resectional arthroplasty for sepsis in one shoulder each. In a more recent article by the same author this Fig.ure did not significantly change at an eleven year end point. (Torchia Cofield and Settergren 1994-1995) In 1989 Brenner and colleagues (Brenner Ferlic Clayton et al. 1989) analyzed the results of fifty-three unconstrained total shoulder arthroplasties using the Kaplan-Meier survivorship curve. Employing a more rigid definition that considered failure as not only the need for reoperation but also patient dissatisfaction with the degree of pain relief the authors reported an eleven year survival of only 73 percent.
In a large multi-center prospective study involving more than 470 unconstrained total shoulder arthroplasties the five year survival was estimated at 97 percent (95 percent confidence interval). (Rockwood 1990) A more rigid definition of failure similar to the criteria proposed by Brenner et al (Brenner Ferlic Clayton et al. 1989) was applied to a subset of these patients whose diagnosis was restricted to osteoarthritis. For these patients failure was defined by one of two parameters. The first parameter as with the two previous studies simply involved the need for reoperation following the index procedure. The second parameter was based upon a patient self-assessment visual analog scale for pain. For this analysis failure was defined as the point in time at which the patient reported shoulder pain which was equal or worse than the preoperative condition. For the osteoarthritis subgroup the probability of five year survival was 92 percent using the more stringent criteria.
Patient self-assessment
As a practical approach to effectiveness measurement which can easily be applied in an active practice one of us (FAM) has used patient self-assessment methods. Since January 1992 all new shoulder patients have been asked to complete the Simple Shoulder Test (SST) (Lippitt Harryman and Matsen 1993; Matsen Lippitt Sidles et al. 1994) to define their pretreatment shoulder function and the SF 36 to characterize there overall health status. The results from these pre treatment questionnaires serve as the baseline on "INGO" for evaluating treatment effectiveness from the perspective of the patient. Followup questions questionnaires reveal the "outcome" of treatment from the patients' perspective. The difference between the outcome and the ingo is the effectiveness of the treatment. Table 16-25 indicates some of these early data for the effectiveness of total or hemi arthroplasty for each of the indicated diagnoses. The preoperative scores are shown to the left of each arrow and the followup score to the right. The data include the SF 36 parameters for total body physical role function and comfort as well as the 12 SST parameters which are specific to the shoulder. The SF 36 scores are the average of the scores of the patients. The SST scores are the percent of the patients answering yes to the indicated question. While some of these numbers are too small for rigorous comparison, in the future, data such as these will indicate the absolute and relative effectiveness of different treatments for different types of glenohumeral arthritis. Dividing the effectiveness by the cost of treatment will reveal the cost effectiveness for different diagnoses by different programs.
Results
Hemiarthroplasty results
The results for the Neer design of hemiarthroplasty have been reported for osteonecrosis osteoarthritis rheumatoid arthritis and the residuals of trauma. When this procedure is applied to the treatment of proximal humeral osteonecrosis the pain relief has been quite good ranging from 91 to 100 per cent and the range of motion of the shoulder approaches normal. When this operation is applied to patients with rheumatoid arthritis osteoarthritis or the residuals of trauma satisfactory pain relief is less consistently achieved but with the exception of 3 of the 11 reported series is still quite acceptable. Range of motion in these latter patients tends to be less and is variable from series to series; average active abduction ranged from one-third to three-quarters normal.
Total shoulder arthroplasty results
The most commonly used total shoulder arthroplasty has been the Neer design. Most patient series contain a mixed diagnostic grouping including patients with rheumatoid arthritis, osteoarthritis, old trauma, and a variety of less common diagnostic categories. As can be seen from the table, the percentage of patients achieving satisfactory pain relief is quite high and quite typically slightly greater than 90% of patients report no or only slight pain following surgery. Motion data following surgery have not been as consistently reported as one might desire but the amount of motion regained seems variable and dependent on diagnostic category. For example in the series reported by Cofield the mean active abduction following surgery for the entire group of patients reported was 120 degrees. (Cofield 1984) The average return of active abduction varied greatly according to diagnosis: 141 degrees for osteoarthritis, 109 degrees for those with post-traumatic arthritis, and 103 degrees for patients with rheumatoid arthritis. The return of movement in Cofield's series was not only dependent on diagnosis but was also highly dependent on the condition of the rotator cuff and shoulder capsule and on the avoidance of complications. (Cofield 1984)
The largest series of total shoulder arthroplasties of this category has been reported by Neer. (Neer Watson and Stanton 1982) He has suggested two systems for grading results. Patients who received a full rehabilitation program were graded as excellent satisfactory or unsatisfactory. To achieve an excellent result the patient was enthusiastic about the operation had no significant pain could use the arm without limitations strength approached normal active elevation of the arm was within 35 degrees of the opposite normal side and external rotation was 90 per cent of the normal side. In patients with a satisfactory result there was no more than occasional pain or aching with weather changes good use of the shoulder for daily activities elevation of at least 90 degrees and rotation to 50% of the normal side. Muscle strength was at least 30% of the normal side and the patients expressed satisfaction with the operation. In an unsatisfactory result the above criteria were not achieved. Neer has suggested a separate evaluation category for patients who have total shoulder replacement but whose muscles could be classified as detached and not capable of recovering function after repair because of fixed contracture or denervation. Patients with substantial bone loss particularly bone loss in the proximal humerus might also be included within this evaluative category. In this setting rehabilitation is aimed at achieving limited goals the purpose being to gain a lesser range of motion but maintain stability. Neer has suggested that this limited-goals rehabilitation is successful when patients with these muscle or bone deficiencies achieve 90 degrees of elevation and 20 degrees of external rotation, maintain reasonable stability, and achieve satisfactory pain relief. Other series of results with this type of prosthesis have been reported. (Weiss Adams Moore et al. 1990)
All series report lucent lines or lucent zones at the glenoid-bone cement junction. These vary considerably in frequency among the different series ranging from 30% to 93% of shoulders reported. The keel portion of this implant serves as the significant means of attachment to the scapula and the lucent zones seen at the cement-bone junction surrounding the keel are of great concern. The median percentage of the number of shoulders analyzed in which a lucent line was identified at the bone-cement junction of the keel part of the component is 36. The argument has been presented that when these lucent lines or zones are seen in patients they are almost always present immediately postoperatively and clearly represent an error in surgical technique. (Neer Watson and Stanton 1982) This may be the most common sequence of events associated with roentgenographic lucent zones at the glenoid bone-cement junction and speaks for the need for meticulous preparation of the bony bed and cementing at the time of surgery. However it has also been reported that these lucent zones have not been present immediately after surgery but rather have developed over time. (Cofield 1984) Green and Norris(Green and Norris 1994b) and Slawson et al (Slawson Everson and Craig 1995) have recently provided a review of imaging techniques for evaluating glenohumeral arthroplasty.
Franklin and co-authors have suggested a classification system for describing the radiographic appearance of the glenoid component. (Franklin Barrett Jackins et al. 1988) In Class 0 there is no lucency. In Class 1 there is lucency at the superior or inferior flange only. In Class 2 there is incomplete lucency at the keel. In Class 3 there is complete lucency up to 2 mm around the component. In Class 4 there is complete lucency greater than 2 mm around the component. In Class 5A the component has translated tipped or shifted in position. And in Class 5B the component has become dislocated from the bone.
In the series by Barrett (Barrett Franklin Jackins et al. 1987) and Cofield (Cofield 1984) analyses have also included a shift in glenoid component position relative to the position achieved immediately following surgery. Analysis of component movement relative to the bone requires the viewing of sequential x-rays over time because often a lucent zone is not seen. This finding implies component loosening but it can easily be overlooked if serial x-rays are not studied.
Management of Glenohumeral Arthritis Image Gallery
Click on any image below to enlarge.